Articles
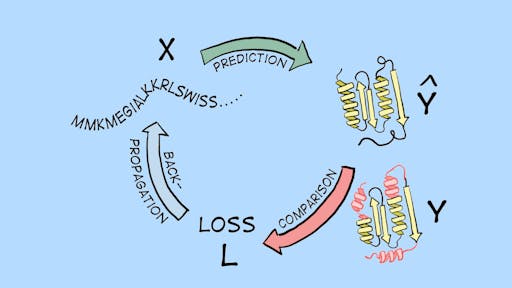
Machine Learning for Protein Engineering - here to stay
Kieran Didi
Engineering T cell receptors for effective T cell activation and immunotherapy
Chris Szeto

AlphaFold – changing the landscape of protein structure prediction
Daniel Williams
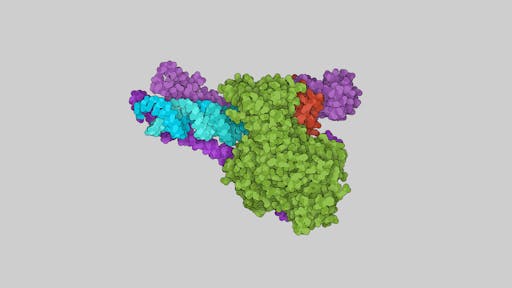
COVID-19 Drugs: Inhibiting viral replication and transcription
Ashley Buckle
SARS-CoV-2: structures light the path to vaccines and treatment
Ashley Buckle

SARS-CoV-2: getting the data out there, and getting more from it
Ashley Buckle
Structural Biology of SARS-CoV-2
Ashley Buckle

It's a small world
Ashley Buckle